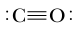11. Bonding & Molecular Structure
Formal Charge
11. Bonding & Molecular Structure
Formal Charge - Video Tutorials & Practice Problems

Get help from an AI Tutor
Ask a question to get started.
Formal Charge is the charge given to an element within a molecule when assuming equal sharing of electrons between bonded elements.
Formal Charge
1
concept
Formal Charge
Video duration:
1mPlay a video:

Net Charge is the sum of individual formal charges within a compound.
2
example
Formal Charge Example 1
Video duration:
56sPlay a video:
3
Problem
ProblemCalculate the formal charges for each of the oxygen atoms within the nitrite ion, NO2–.

A
0, -1
B
+1, 0
C
-1, -1
D
+1, +1
4
Problem
ProblemCalculate the formal charge of the carbon atom within a carbon monoxide molecule.
a) –1
b) +2
c) 0
d) +
e) –2
A
–1
B
+2
C
0
D
+1
E
–2
5
Problem
ProblemBased on calculated formal charges, determine the overall charge (net charge) for the following compound.
a) –3
b) +4
c) +1
d) –1
e) +2
A
–3
B
+4
C
+1
D
–1
E
+2
6
Problem
ProblemWhich element within the thiocyanate ion possesses a negative charge?
a) C
b) S
c) N
d) All of them
e) None of them
A
C
B
S
C
N
D
All of them.
E
None of them.
Do you want more practice?
We have more practice problems on Formal Charge
Additional resources for Formal Charge
PRACTICE PROBLEMS AND ACTIVITIES (12)
- In the Lewis structure shown here, A, D, E, Q, X, and Z represent elements in the first two rows of the perio...
- Consider the Lewis structure for the polyatomic oxyanion shown here, where X is an element from the third per...
- Which is the best electron-dot structure for the thiosulfate ion (S2O3 2-) based on the rules of formal charge...
- Which one of these statements about formal charge is true? (a) Formal charge is the same as oxidation number. ...
- In the vapor phase, BeCl2 exists as a discrete molecule. (c) On the basis of the formal charges, which Lewis s...
- Consider the Lewis structure shown below. (a) Does the Lewis structure depict a neutral molecule or an ion? I...
- Use formal charges to identify the better Lewis structure.
- In N2O, nitrogen is the central atom and the oxygen atom is terminal. In OF2, however, oxygen is the central a...
- What are the formal charges of the atoms shown in red?
- What are the formal charges of the atoms shown in red?
- The following three Lewis structures can be drawn for N2O: (a) Using formal charges, which of these three res...
- Calculate the formal charge on the chlorine (cl) atom.